Who We Are & Why We’re Here
25+ YEARS OF INNOVATION
Est. 1998
For more than a decade, we’ve partnered with funded startups, helping them transform innovative medical device concepts into market-ready solutions, guided by an integrated team and a proven, end-to-end development approach.
YOUR VISION, OUR EXPERTISE
ISO 13485:2016 Certified Quality System
Biotex delivers value by maintaining certified, validated, and industry-recognized capabilities, allowing us to provide premium products and services from initial design through successful commercialization. Our commitment to quality, safety, and best practices will be your leverage.
IEC 17025 Accredited Lab
Streamline your operations by tapping into our in-house testing services. Criterion, our ISO/IEC 17025–certified testing laboratory, supports your path to medical device compliance. We help you meet safety and regulatory requirements with confidence, a critical phase in market readiness.
DEEP IN THE HEART OF TEXAS
Located in the heart of Houston, we are a short distance from the world’s largest Medical Center. We are accessible, equipped with a spacious office, in-house labs and maker space, an ISO Class 7 clean room, and a production warehouse, ready to accommodate your needs.

Built for Innovators
Biotex has incubated, accelerated, spun-out and launched companies, navigated regulatory approvals, led
clinical trials, and scaled manufacturing, demonstrating our ability to bring significant value to our
partners for the entire product journey.
Our extensive experience and expertise in developing medical technologies do not just fuel our commitment to
excellence. They're the backbone of our being.
At Biotex, our business approach defines who we are - rigorous, collaborative, passionate, and driven to
shape the future of healthcare and MedTech.
-
For Inventors
You understand the gaps in care.
We transform your insights into safe, functional, and clinically viable devices designed to address pain points, patient needs, and practical use in clinical settings. -
For Established Companies
From early prototypes to market-ready products and scaled manufacturing, our integrated services can fill the gaps at any stage of development.
We work to support and complement your team, enabling seamless, strategic collaboration. -
For Investors
A trusted partner for early-stage MedTech, we bring decades of experience guiding devices from ideation to exit.
We help accelerate your path to exit through deep technical and business development expertise, access to our state-of-the-art facility, and a dedicated team committed to your success.

Our Mission
-
Fuel Innovation
We utilize modern, cutting-edge technologies to solve life sciences problems.
-
Revolutionize Healthcare
Our commitment is to develop superior medical devices that positively impact human lives. We utilize state-of-the-art technologies, collaborate with industry experts, and support the full device journey from concept to commercialization.
-
Create Life-Changing Devices
We value science and human connection, allowing us to pair evidence-based thinking with a strong understanding of patient needs as we implement our value-driven business model and create groundbreaking, transformative medical solutions.
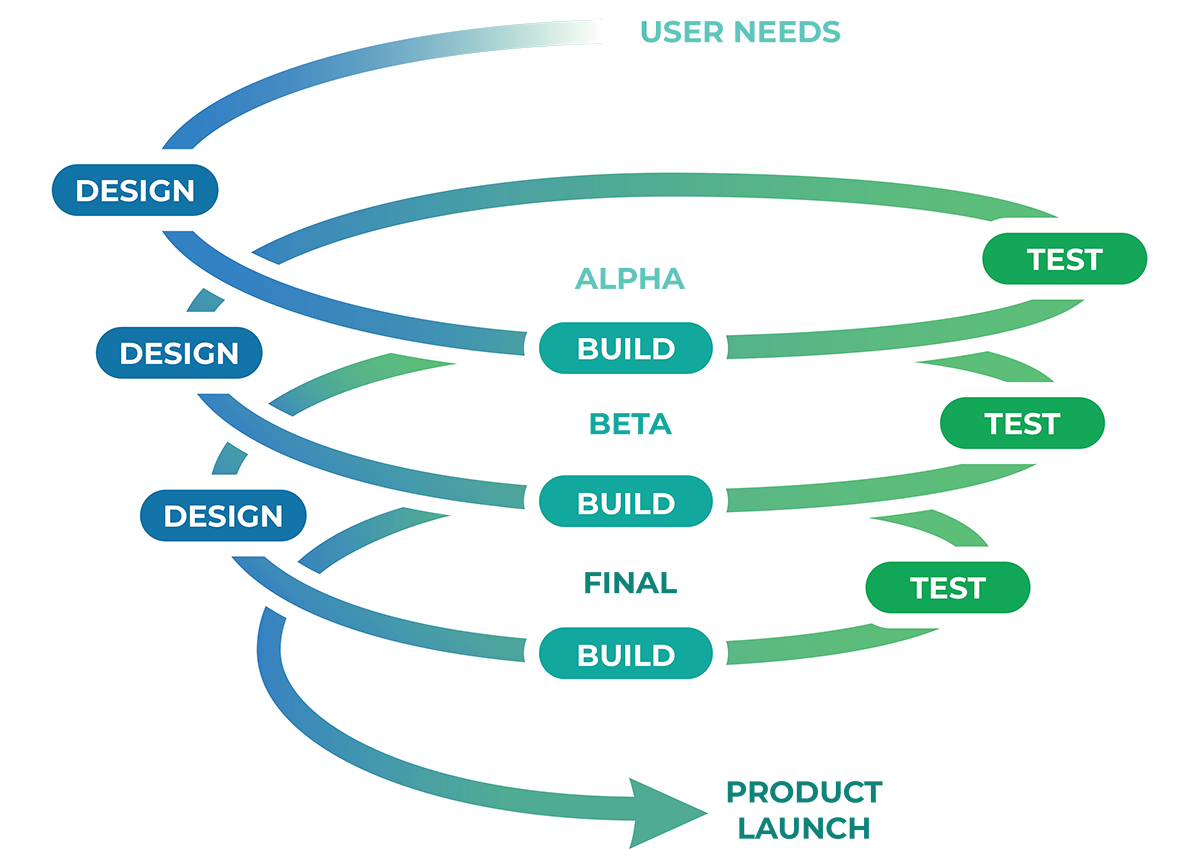
Navigating the Full Product Journey
We know the path because we've navigated it repeatedly, not once but over two decades.
Bringing a medical device to market demands careful navigation through design,
testing, pre-clinical/clinical studies, regulatory clearance/approval, manufacturing, and order
fulfillment.
Biotex offers an integrated development environment that keeps every phase aligned. With our
multidisciplinary teams under one roof, programs move efficiently from early concept through
clinical
readiness and scalable production.
The HELIX Model
Cross-functional teams working in parallel across every phase.
Our integrated teams operate in parallel across the full development lifecycle,
with engineering, testing, clinical, regulatory, and manufacturing working together from start to
finish.
The Helix model consolidates feedback from every department from the onset, preventing silos,
reducing rework, and keeping our projects moving without interruption or delay.